MedTech Compliance Articles
Expert guides on EU MDR, Clinical Evaluation, and Literature Review strategies.

CiteMed | EU MDR & Clinical Evaluation Insights
In-depth articles and resources on EU MDR, IVDR, and clinical evaluation for regulatory affairs professionals.
Latest Articles
In Vitro Diagnostic Regulation (IVDR) Device Risk Management Challenges
All medical devices have their merit since all of [...]
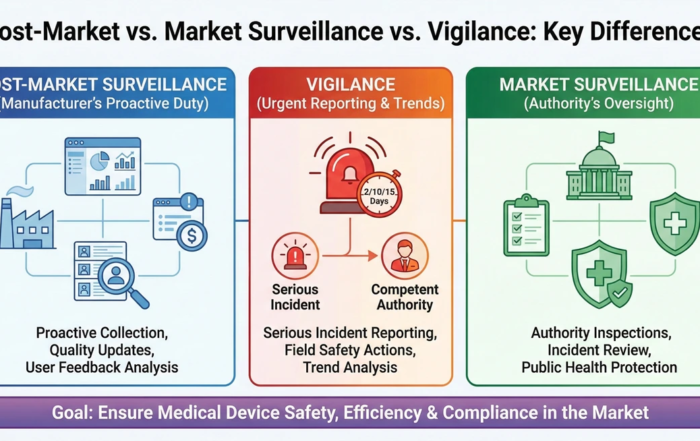
Post-Market Surveillance, Market Surveillance, and Vigilance – What is the Difference?
Most regulatory staff can define post-market surveillance relatively succinctly. [...]
Medical Device Regulations – Links You Should Be Aware Of
Medical device regulations are no joke. It's pretty important [...]
Literature Search: How Much Research is Enough?
The research process is often thought of as a linear, [...]
The Top 5 Regulatory Requirements for the Medical Device Industry
Being in the regulatory industry is not as cut [...]
Vigilance Reporting Under the MDR: Insider’s Guide
Implementing the European Medical Devices Regulation (MDR; 2017/745) and [...]