Systematic Literature Review Course
Welcome to the Systematic Literature Review Course!

Unexpected Documents You Might Need for Your Clinical Evaluation Report
Medical device regulation is important. Not just because we work with them. But because they control and regulate devices [...]
Software as a Medical Device
Healthcare is undergoing a paradigm shift as cutting-edge technology makes its way into medical devices, tele-health, and digital health [...]
Quality Management System: What ISO Standards Say About QMS
A quality management system is a huge part of producing medical devices. Whether you like the process or not, [...]
In Vitro Diagnostic Regulation (IVDR) Device Risk Management Challenges
All medical devices have their merit since all of them are used in the medical field. However, the In [...]
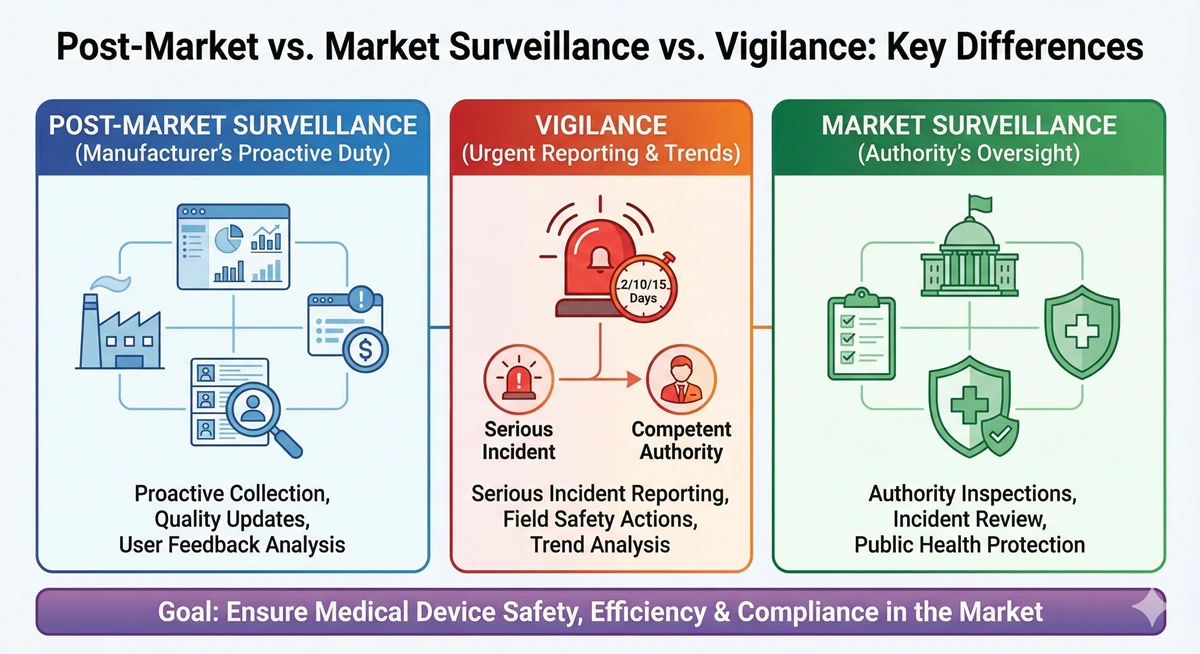
Post-Market Surveillance, Market Surveillance, and Vigilance – What is the Difference?
Most regulatory staff can define post-market surveillance relatively succinctly. Perhaps also vigilance. But when it comes to market surveillance, [...]
Medical Device Regulations – Links You Should Be Aware Of
Medical device regulations are no joke. It's pretty important to get the regulations right, since even a single missed [...]
Literature Search: How Much Research is Enough?
The research process is often thought of as a linear, straightforward path. You begin with a question and then proceed [...]
The Top 5 Regulatory Requirements for the Medical Device Industry
Being in the regulatory industry is not as cut and dry as most people think. Sure, you do have [...]
Vigilance Reporting Under the MDR: Insider’s Guide
Implementing the European Medical Devices Regulation (MDR; 2017/745) and the In Vitro Diagnostic Medical Devices Regulation (IVDR; 2017/746) have [...]