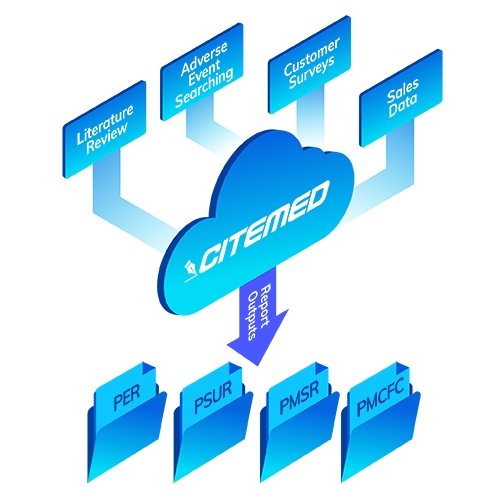
Comprehensive Reporting
The Only Post Market Surveillance Decision You Need to Make
Post Market Surveillance under MDR has become a pain. Believe us, we understand. With multiple documents to juggle, different submission intervals, and (*cough*) ambiguity in the regs, it’s hard to stay organized.
Our team has built the experience, tools and processes to handle it all, year after year. All you have to do let us use them.

Featured In
Our Clients Say
Free Articles and Post Market Surveillance Whitepapers
Post-Market Surveillance Under EU MDR: Building a Proactive PMS System
Master post-market surveillance under EU MDR to ensure compliance and success. Optimize your strategies and enhance product safety effectively today.
MDR Literature Review Software: Automated Reviews for EU MDR and IVDR Compliance
Streamline your compliance process with effective MDR literature review software. Enhance efficiency and ensure accurate reviews for better results today.
EU MDR Consultant Selection: Essential Criteria to Consider
Choosing the right EU MDR consultant is vital for your compliance success. Learn the key criteria to consider for effective selection and a smooth process.
Worry-Free EU MDR Compliance
All Your Vigilance Data In One Place

If You Are:
Short on staff and resources to conduct multiple Vigilance reports every quarter.
Perfectionists that want a consistent system and operating procedure to satisfy Post Market Surveillance requirements.
In need of a single centralized platform to securly store your AE Reports and trending data over time.