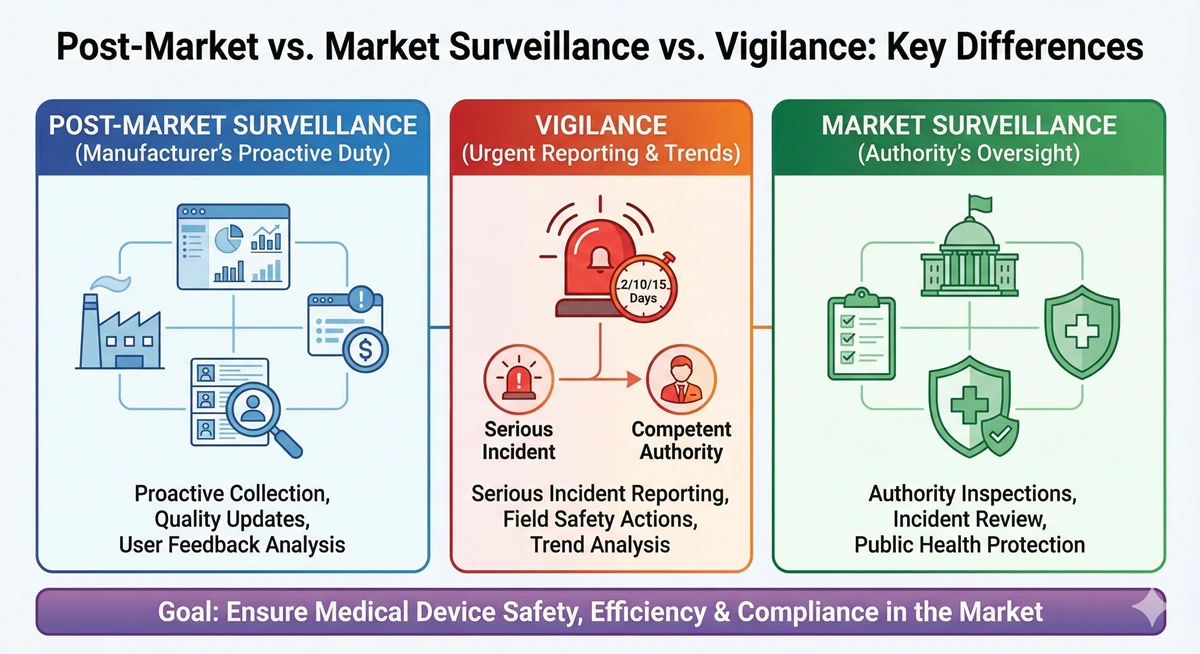
MedTech Compliance Articles
Expert guides on EU MDR, Clinical Evaluation, and Literature Review strategies.

CiteMed | EU MDR & Clinical Evaluation Insights
In-depth articles and resources on EU MDR, IVDR, and clinical evaluation for regulatory affairs professionals.
Latest Articles
Five Practical Tips for Writing a Compliant Clinical Evaluation Report (CER)
Even for experienced medical writers and regulatory staff, writing [...]
EU In Vitro Diagnostic Regulation (IVDR): Frequently Asked Questions
The regulatory landscape in Europe is changing. In 2021, [...]
Analyzing the Differences between CERs for MDR versus MDD
Implementing the MDR or Medical Device Regulation will bring [...]
IVDR: State-of-the-Art for In Vitro Diagnostic Medical Devices
With the updates to the European Medical Device and [...]
Publishing Clinical Trials Results in Journals
Clinical trials are a crucial piece of launching a [...]
Clinical Trials Reporting Requirements in EU Countries
Clinical trials are important. The discovery of any new [...]